 
That is, the state of a natural system itself can be reversed, but not without increasing the entropy of the system's surroundings, that is, both the state of the system plus the state of its surroundings cannot be together, fully reversed, without implying the destruction of entropy.įor example, when a path for conduction or radiation is made available, heat always flows spontaneously from a hotter to a colder body. It asserts that a natural process runs only in one sense, and is not reversible. The second law is concerned with the direction of natural processes. Conceptually, the first law describes the fundamental principle that systems do not consume or 'use up' energy, that energy is neither created nor destroyed, but is simply converted from one form to another. It can be linked to the law of conservation of energy. The first law of thermodynamics provides the definition of the internal energy of a thermodynamic system, and expresses its change for a closed system in terms of work and heat. Introduction Heat flowing from hot water to cold water The second law of thermodynamics allows the definition of the concept of thermodynamic temperature, but this has been formally delegated to the zeroth law of thermodynamics. The first rigorous definition of the second law based on the concept of entropy came from German scientist Rudolf Clausius in the 1850s and included his statement that heat can never pass from a colder to a warmer body without some other change, connected therewith, occurring at the same time. Its first formulation, which preceded the proper definition of entropy and was based on caloric theory, is Carnot's theorem, formulated by the French scientist Sadi Carnot, who in 1824 showed that the efficiency of conversion of heat to work in a heat engine has an upper limit. 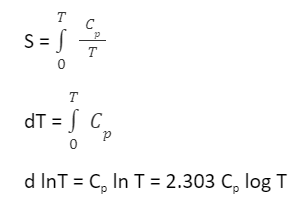
The second law has been expressed in many ways. Statistical mechanics provides a microscopic explanation of the law in terms of probability distributions of the states of large assemblies of atoms or molecules. Historically, the second law was an empirical finding that was accepted as an axiom of thermodynamic theory. An increase in the combined entropy of system and surroundings accounts for the irreversibility of natural processes, often referred to in the concept of the arrow of time. The second law may be formulated by the observation that the entropy of isolated systems left to spontaneous evolution cannot decrease, as they always tend toward a state of thermodynamic equilibrium where the entropy is highest at the given internal energy. For example, the first law allows the process of a cup falling off a table and breaking on the floor, as well as allowing the reverse process of the cup fragments coming back together and 'jumping' back onto the table, while the second law allows the former and denies the latter. It predicts whether processes are forbidden despite obeying the requirement of conservation of energy as expressed in the first law of thermodynamics and provides necessary criteria for spontaneous processes. The second law of thermodynamics establishes the concept of entropy as a physical property of a thermodynamic system. Another statement is: "Not all heat can be converted into work in a cyclic process." A simple statement of the law is that heat always flows spontaneously from hotter to colder regions of matter (or 'downhill' in terms of the temperature gradient). It also covers the labor required to convert energy from one form to another.The second law of thermodynamics is a physical law based on universal empirical observation concerning heat and energy interconversions. Thermodynamics is the study of the energy changes that occur as a result of temperature and heat variations. Penetration Testing Interview Questions.Software Engineering Interview Questions. 
Top 10 System Design Interview Questions and Answers.Java Multithreading Interview Questions. 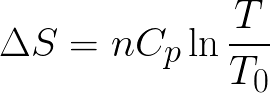
Food delivery system using HTML and CSS.Building a Survey Form using HTML and CSS.Top 20 Puzzles Commonly Asked During SDE Interviews.Top 100 DSA Interview Questions Topic-wise. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
